Visby brings point-of-care PCR to the deep south with an appearance at NACCHO360.
Author: Jed Royer
Diagnosing covid-19: Barriers to pandemic planning
We don’t know. The symptoms of the disease are nonspecific, and many people are not even aware they carry the virus. In fact, the CDC estimates that 40% of people with coronavirus symptoms show no symptoms, with other researchers suggesting the rate may be even higher.
Visby Medical Appoints Industry Veteran John W. Kuo as Chief Legal Officer
Visby Medical™, a leading medical diagnostic company, today announced the appointment of John W. Kuo as the company’s new Chief Legal Officer. An experienced veteran in corporate law, Kuo brings more than 30 years of experience across multiple industries, including nearly 20 years in the healthcare industry.
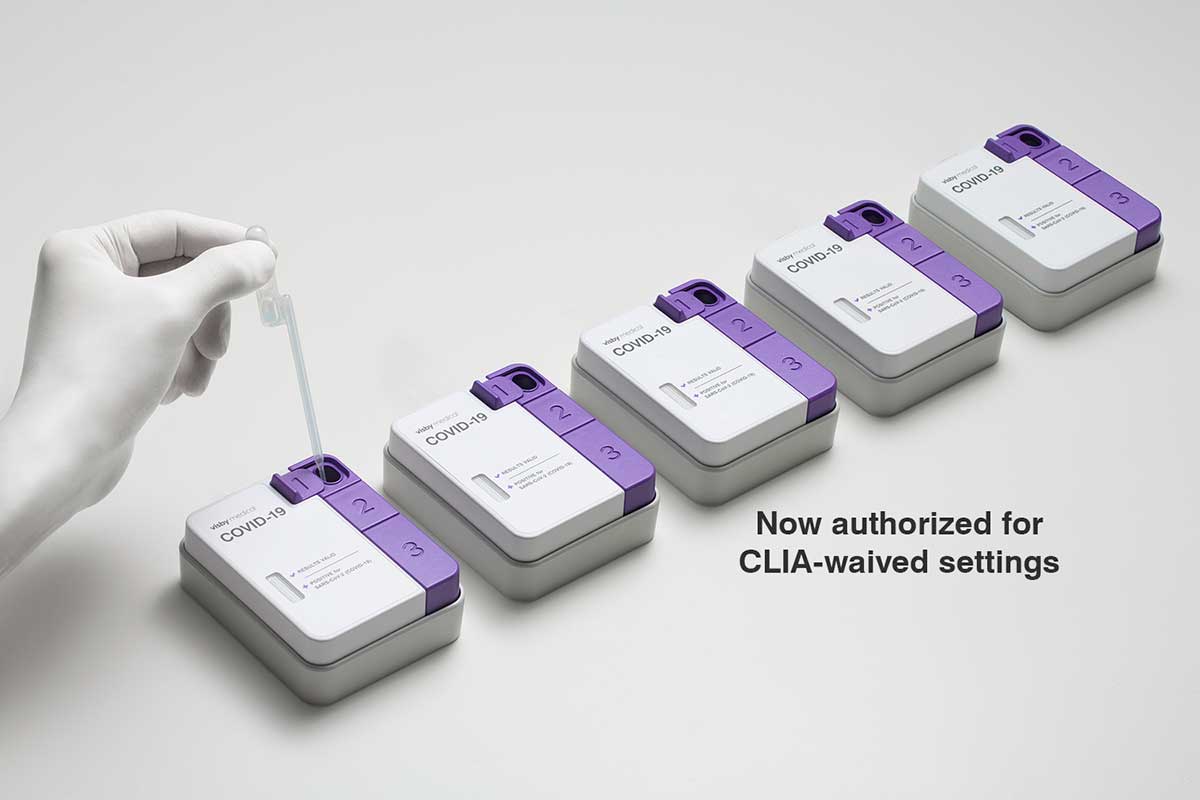
Visby Medical’s COVID-19 PCR Point of Care Test Authorized for Use in CLIA Waived Settings
Expanding critical access to the only single-use, rapid PCR test, at the point of care.
Visby Medical speeds development of a rapid Flu-COVID PCR test designed for at-home use with BARDA funding
Visby Medical pairs speed with the accuracy of gold-standard PCR testing in a single-use device, so doctors don’t have to fight blind.
Lancet publishes paper on the Visby Medical point-of-care PCR technology
A study of the performance of the Visby Medical Sexual Health Test, a single-use, point-of-care PCR device.
What is PCR?
PCR’s superpower lies in its super sensitivity, which enables it to sniff out even low levels of a pathogen in minutes. Here’s how it works.
Visby Medical’s Personal PCR Receives Funding from National Institute of Health, RADx Program
Visby Medical, an infectious disease diagnostic company, received funding from the National Institutes of Health as part of the organization’s Rapid Acceleration of Diagnostics (RADx) program, an initiative that aims to combat SARS-CoV-2 and increase national testing capacity for COVID-19.
Personal PCR: Rapid, Reliable, and Prolific
Personal PCR could one day become as common as the common cold. In the future, the device will deliver lab accurate results anywhere and to anyone — forever changing our relationship with diagnostics.
Visby Medical’s Personal PCR Device Receives FDA Emergency Use Authorization for Moderate-Complexity Lab Environments
Seven-year-old startup emerges from stealth mode, provides adaptable and rapid testing technology to frontline lab personnel fighting the pandemic